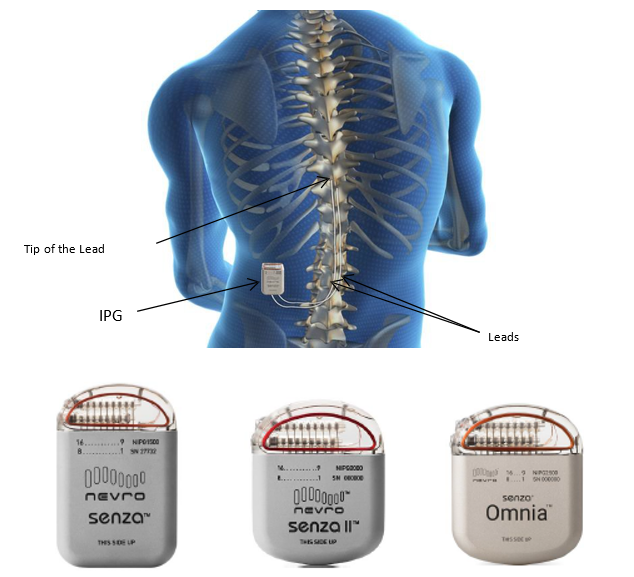
Nevro receives CE mark for full-body MRI conditional labelling with the Senza system - NeuroNews International

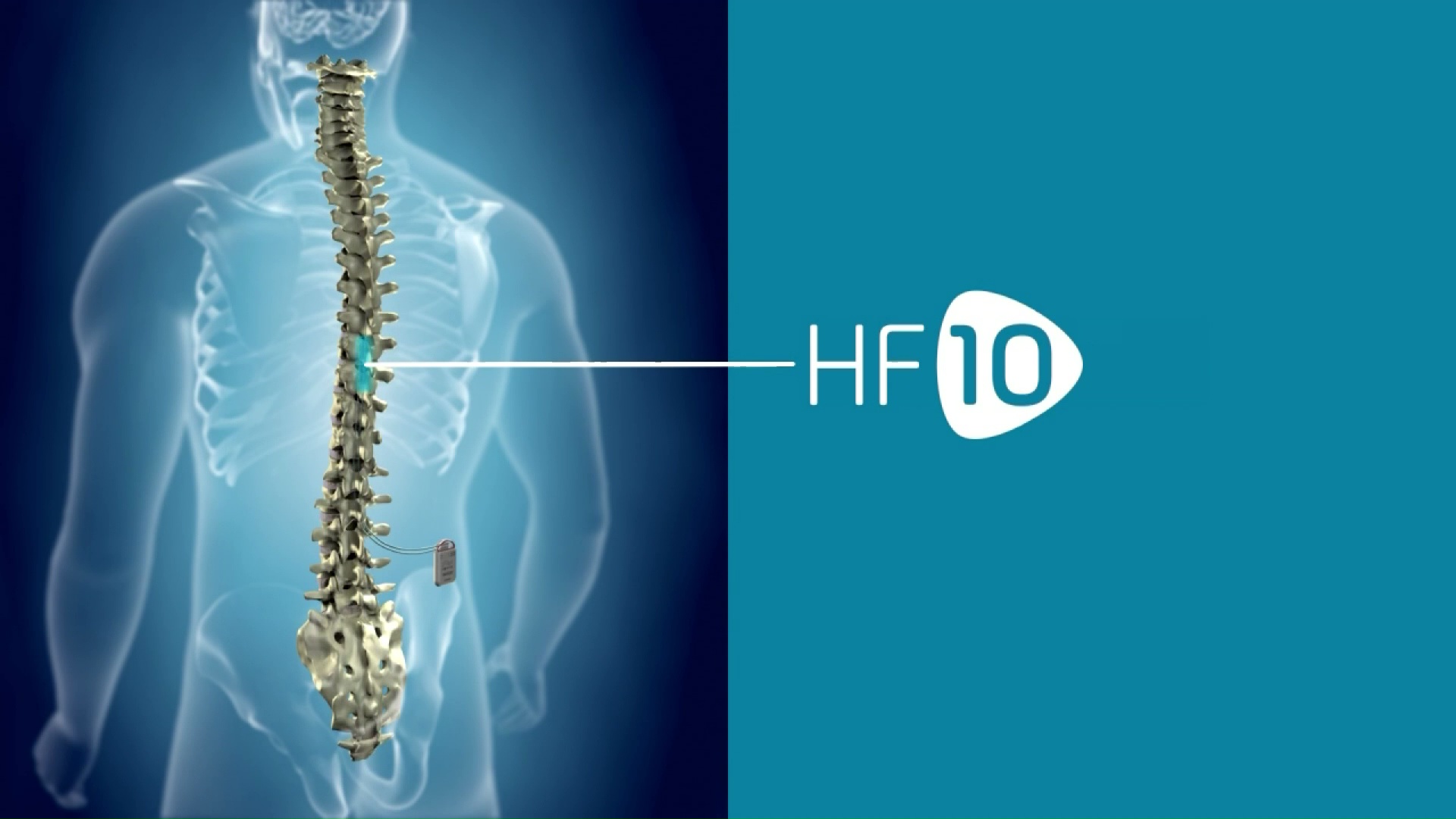
Nevro Receives FDA Approval For Senza II Spinal Cord Stimulation System Delivering HF10 Therapy | Orthopedic Design Technology

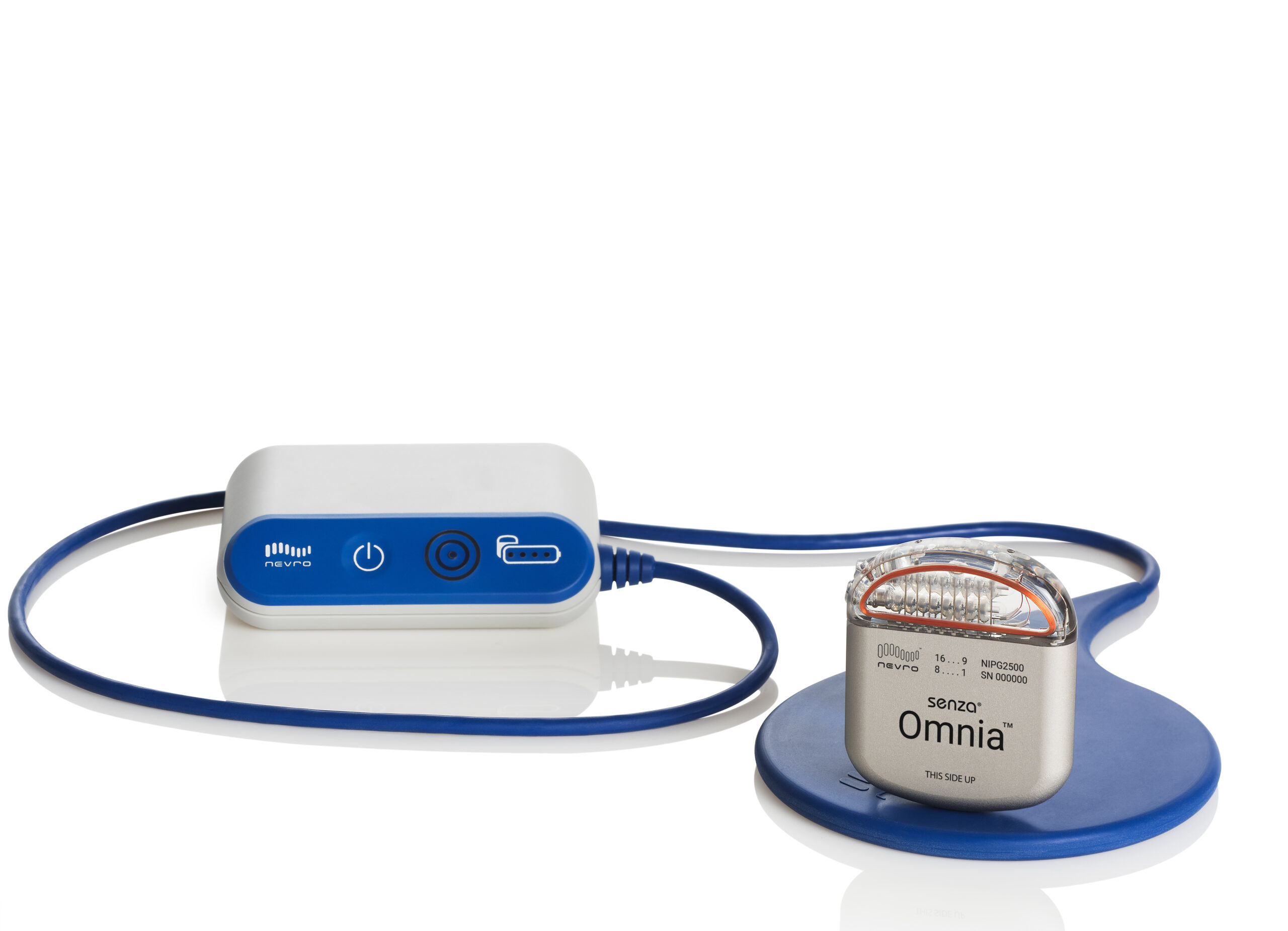
Nevro Corp. - Nevro Announces FDA Approval of HFX iQ™ Spinal Cord Stimulation System to Personalize the Treatment of Chronic Pain

Nevro Announces FDA Approval of its 10 kHz High Frequency Spinal Cord Stimulation Therapy for Treatment of Chronic Pain Associated with Painful Diabetic Neuropathy (PDN)

Nevro Corp. - Nevro Announces U.S. Full Market Launch of Revolutionary HFX iQ™ Spinal Cord Stimulation System to Personalize Treatment of Chronic Pain

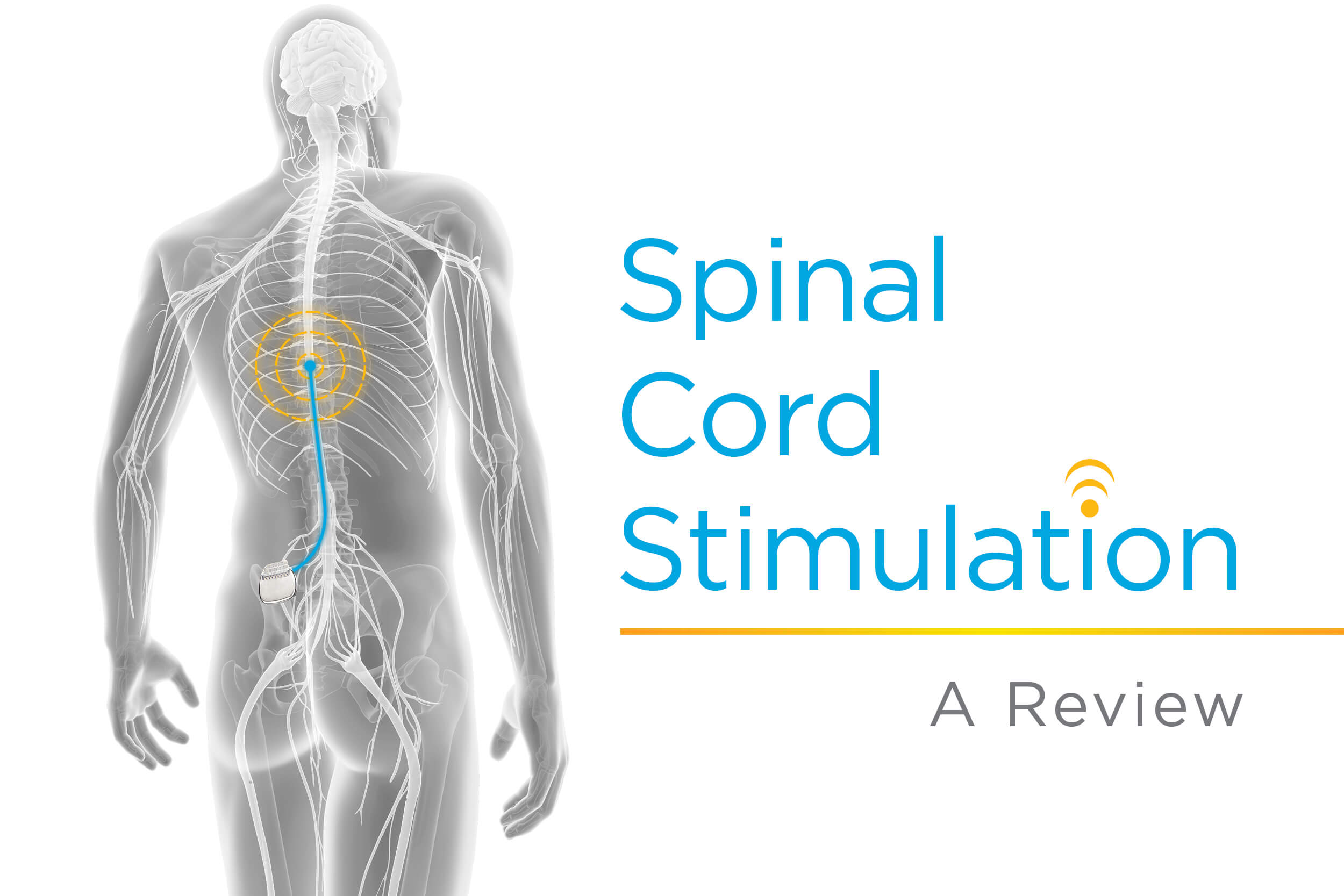
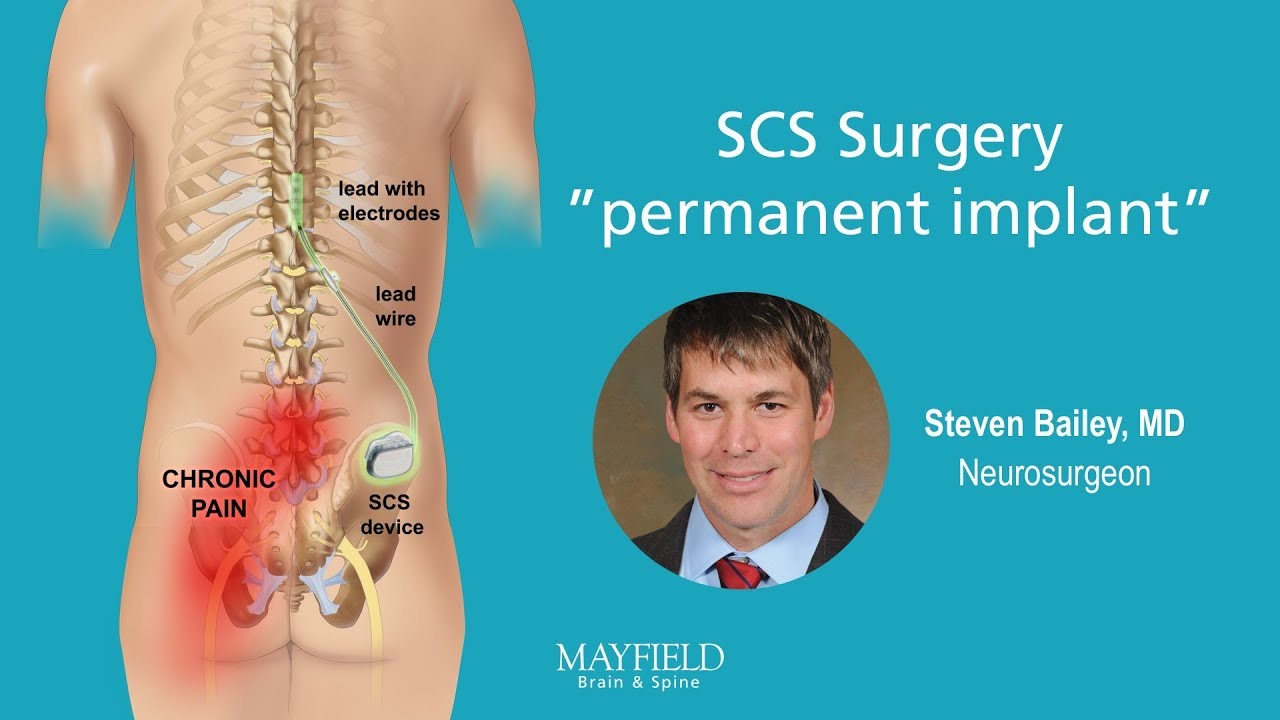
Multicentre, double-blind, randomised, sham-controlled trial of 10 khz high-frequency spinal cord stimulation for chronic neuropathic low back pain (MODULATE-LBP): a trial protocol | Trials | Full Text